
























Friedel-Crafts Alkylation of Benzene
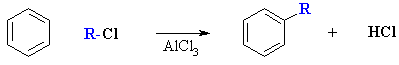
Summary.
- Overall transformation : Ar-H to Ar-R
- Named after Friedel and Crafts who discovered the reaction in 1877.
- Reagent : normally the alkyl halide (e.g. R-Br or R-Cl) with aluminum trichloride, AlCl3, a Lewis acid catalyst
- The AlCl3 enhances the electrophilicity of the alkyl halide by complexing with the halide
- Electrophilic species : the carbocation (i.e. R +) formed by the "removal" of the halide by the Lewis acid catalyst
- The reactive electrophile, the carbocation is prone to rearrangement to a more stable carbocation which will then undergo the alkylation reaction.
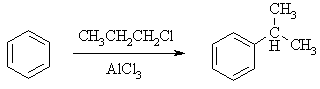
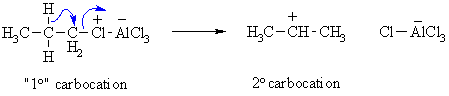
- Friedel-Crafts reactions are limited to arenes as or more reactive than mono-halobenzenes
- Other Lewis acids such as BF3, FeCl3 or ZnCl2 can also be used
- Other sources of carbocations can also be used:
- from loss of water from alcohols treated with acid
- from the protonation of alkenes by acid
- Alkylation products can also be obtained by the reduction of Friedel-Crafts acylation products
|
|
|
| Step 1: The alkyl halide reacts with the Lewis acid to form a a more electrophilic C, a carbocation |
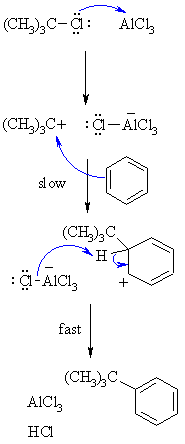 |
| Step 2: The p electrons of the aromatic C=C act as a nucleophile, attacking the electrophilic C+. This step destroys the aromaticity giving the cyclohexadienyl cation intermediate. |
|
| Step 3: Removal of the proton from the sp3 C bearing the alkyl- group reforms the C=C and the aromatic system, generating HCl and regenerating the active catalyst. |
© LEV