






















Alkenes
Family name: suffix: -ene
General formula CnH2n (Where n must be >1)
In contrast to alkanes where the carbon atom will form 4 single bonds (or s (sigma) bonds) to 4 different atoms, a carbon atom of an alkene will form bonds to three other atoms. Two of these bonds will be single bonds while the third is a double bond (which consists of both a s (sigma) bond and a p (pi) bond to the same atom). The alkenes are also referred to as unsaturated hydrocarbons (as they contain multiple bonds). The geometry of these unsaturated or sp2 hybridized carbon atoms is trigonal planar (bond angles of approximately 120o with all four atoms laying in a plane). The general formula for a alkene is CnH2n where n is the number of carbon atoms present in the molecule. The loss of a pair of hydrogen atoms indicates the presence of the double bond between two carbon atoms.The unsaturated hydrocarbons are named using a different suffix to the root name of the hydrocarbon, -ene. Since there must be a minimum of two carbon atoms for a multiple bond to occur, the simplest alkene is ethene.
Comparison of simple alkenes:
|
|
|
|
Properties |
|
|
|
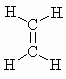 |
m.pt.= 104o b.pt. = 169.44o DHf = +52.3kJ/mol DHc = -1411.0 kJ/mol |
|
|
|
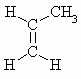 |
m.pt.= 87o b.pt. = 225.45o DHf = +20.4 kJ/mol DHc = -2058 kJ/mol |
| C4H8 | But-1-ene | 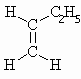 |
m.pt.= 87.8o b.pt. = 266.84oC DHf = -0.1 kJ/mol DHc = -2717.3 kJ/mol |
| C4H8 | cis But-2-ene | 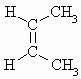 |
m.pt.= 134oC b.pt. = 276.87o DHf = -7.0 kJ/mol DHc = -2710.4 kJ/mol |
| C4H8 | trans But-2-ene | 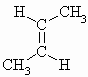 |
m.pt.= 167.6o b.pt. = 274o DHf = -11.9 kJ/mol DHc = -2702.2 kJ/mol |
NOTE: The convension in drawing alkenes is: the double bond is indicated by a pair of parallel lines between the two carbon atoms, and the bond angles should be approximately 120o. Ethene is an unusual molecule in that it is flat like benzene.
The naming of the alkenes is similar to alkanes with the following addition.
- The double bond will be higher in priority than any simple alkyl or halide group, therefore the root name is derived by finding the longest chain (or ring) which contains both atoms of the double bond.
- All other groups will be substituents on this parent chain (or ring).
- The location of the double bond is indicated by the number of the first carbon atom of the double bond, and the bond is given the lowest possible number, i.e. 1-propene not 2-propene. There are two convensions in placing the number: the less common method is to put the number between the root and suffix, i.e. prop-1-ene (example four above); the more common method (and the one we will use exclusively) is to place the number before the root name, i.e. 1-propene. It is common to leave out the number when the double bond is between atoms 1 and 2, i.e. propene is more commonly used than 1-propene.
© LEV