






















The reaction of conc. sulphuric acid with propene
This is an electrophic addition reaction
This is typical of the reaction with unsymmetrical alkenes. An unsymmetrical alkene has different groups at either end of the carbon-carbon double bond.
If sulphuric acid adds to an unsymmetrical alkene like propene, there are two possible ways it could add. You could end up with one of two products depending on which carbon atom the hydrogen attaches itself to.
However, in practice, there is only one major product.

This is in line with Markovnikov's Rule which says:
- When a compound HX is added to an unsymmetrical alkene, the hydrogen becomes attached to the carbon with the most hydrogens attached to it already. In this case, the hydrogen becomes attached to the CH2 group, because the CH2 group has more hydrogens than the CH group. Notice that only the hydrogens directly attached to the carbon atoms at either end of the double bond count. The ones in the CH3 group are totally irrelevant.
|
|
|
| Step
1: An acid / base reaction. Protonation of the alkene to generate the more stable carbocation. The p electrons act as a Lewis base. |
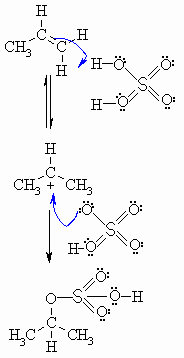 |
| Step
2: Attack of the nucleophilic hydrogen sulphate ion on the electrophilic carbocation creates the initial product. |
|
| Step
3:(not shown) Addition of water diplaces the sulphuric acid forming an alcohol. |
© LEV