






















Acidified Manganate VII
We'll look at the reaction with ethene. Other alkenes react in just the same way.
Manganate(VII) ions are a strong oxidising agent, and in the first instance oxidise ethene to ethandiol (old name: ethylene glycol).
Looking at the equation purely from the point of view of the organic reaction:
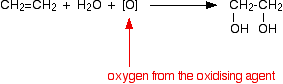
The full equation depends on the conditions.
Under acidic conditions, the manganate(VII) ions are reduced to manganese(II) ions.
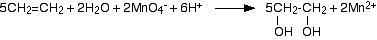
The product, ethandiol, is itself quite easily oxidised by manganate(VII) ions, and so the reaction won't stop at this point unless the potassium manganate(VII) solution is very dilute, very cold, and preferably not under acidic conditions.
That means that this reaction has little use as a way of preparing ethandiol. Its only real use is in testing for carbon-carbon double bonds - and even then it isn't very good!
The situation with acidified potassium manganate(VII) solution is difficult because it has a tendency to break carbon-carbon bonds. It reacts destructively with a large number of organic compounds and is rarely used in organic chemistry.
You could use alkaline potassium manganate(VII) solution if, for example, all you had to do was to find out whether a hydrocarbon was an alkane or an alkene - in other words, if there was nothing else present which could be oxidised.
It isn't a useful test. Bromine water is far more clear cut.