






















Basic IUPAC Organic Nomenclature
E- and Z- nomenclature of alkenes
On the previous pages, we looked at naming alkenes as cis- and trans-.It is important to note that the two methods are different (i.e. they are based on different rules) and they are NOT interchangeable, see below for an example.
The cis- / trans- style is based on the longest chain whereas the E/Z style is based on a set of priority rules.
You need to know both styles if you started studying the syllabus in 2008.
 |
|
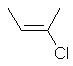
| cis-but-2-ene or (Z)-but-2-ene |
cis-2-chlorobut-2-ene
or (E)-2-chlorobut-2-ene |
The E- and Z- style is more reliable and particularly suited to highly substituted alkenes, especially when the substituents are not alkyl groups.
The Cahn-Ingold-Prelog priority rules are used for naming geometric isomers (e.g. E- or Z-alkenes) and other stereoisomers (see later).
These rules are based on atomic number, and the first point of difference.
- Imagine each alkene as two pieces, each piece containing one of the sp2 C
- Assign the priority (high = 1, low = 2) to each group on each sp2 C based on atomic number
- Determine the relative position of the two higher priority groups
- If they are on the same side then it is a (Z)-alkene (German; zusammen = together)
- If they are on opposite sides then it is an (E)-alkene (German; entgegen = opposite)
- If there is more than
one C=C that can be E/Z, then
the location needs to be included with the locant, e.g. (2E,4Z)-
- Isomer 1 :
 |
 |
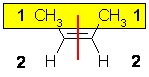 |
| Step 1 : split the
alkene |
Step 2 : assign the
relative priorities |
Step 3 : look at the
relative positions of the higher priority groups : same side = Z, hence
(Z)-but-2-ene. |
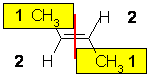
- Isomer
2 :

 |
|
|
The two substituents are -CH3 and H, so since the atomic numbers C > H then the -CH3 group is higher priority. Therefore the two high priority groups are on the opposite side, then this is (E)-but-2-ene. |
|
Subrules:
- Isotopes: H vs D ? Since isotopes have identical atomic numbers, the mass number is used to discriminate them so D > H
- Same atom attached ? By moving out one unit at a time, locate the first point of difference and apply rules there.
© LEV