






















Reaction of Alkenes with halogens.
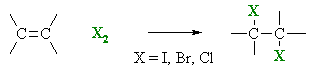
This
reaction occurs rapidly it results in the loss of colour from Iodine
and bromine.
Reaction type: Electrophilic Addition
Summary.
- Overall transformation : C=C to X-C-C-X
- Reagent : normally the halogen (e.g. Br2) in an inert solvent like methylene chloride, CH2Cl2, but can also occur as a gas phase reaction.
- Regioselectivity : not relevant since both new bonds are the same, C-X.
- Reaction proceeds via cyclic halonium ion.
- Stereoselectivity : anti since the two C-X bonds form in separate steps one from Br2 the bromide ion Br-.
|
|
|
| Step
1: The p electrons act as a nucleophile, attacking the bromine, displacing a bromide ion but forming a cationic cyclic bromonium ion as an intermediate. |
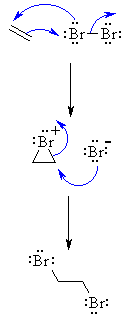 |
| Step
2: Attack of the nucleophilic bromide from the side away from the bromonium center in an SN2 like fashion opens the cyclic bromonium ion to give overall trans addition. |
© LEV